|
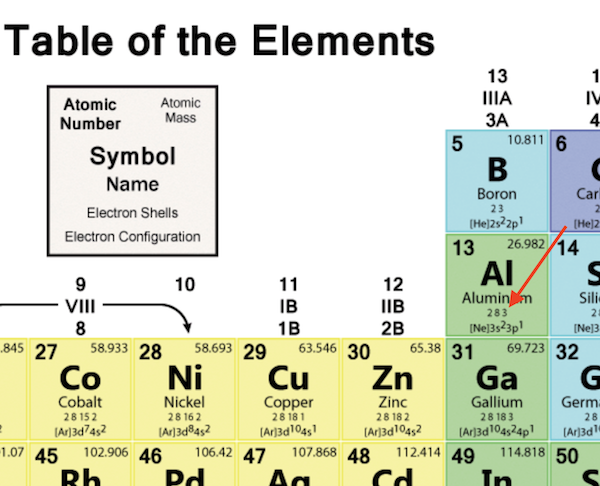
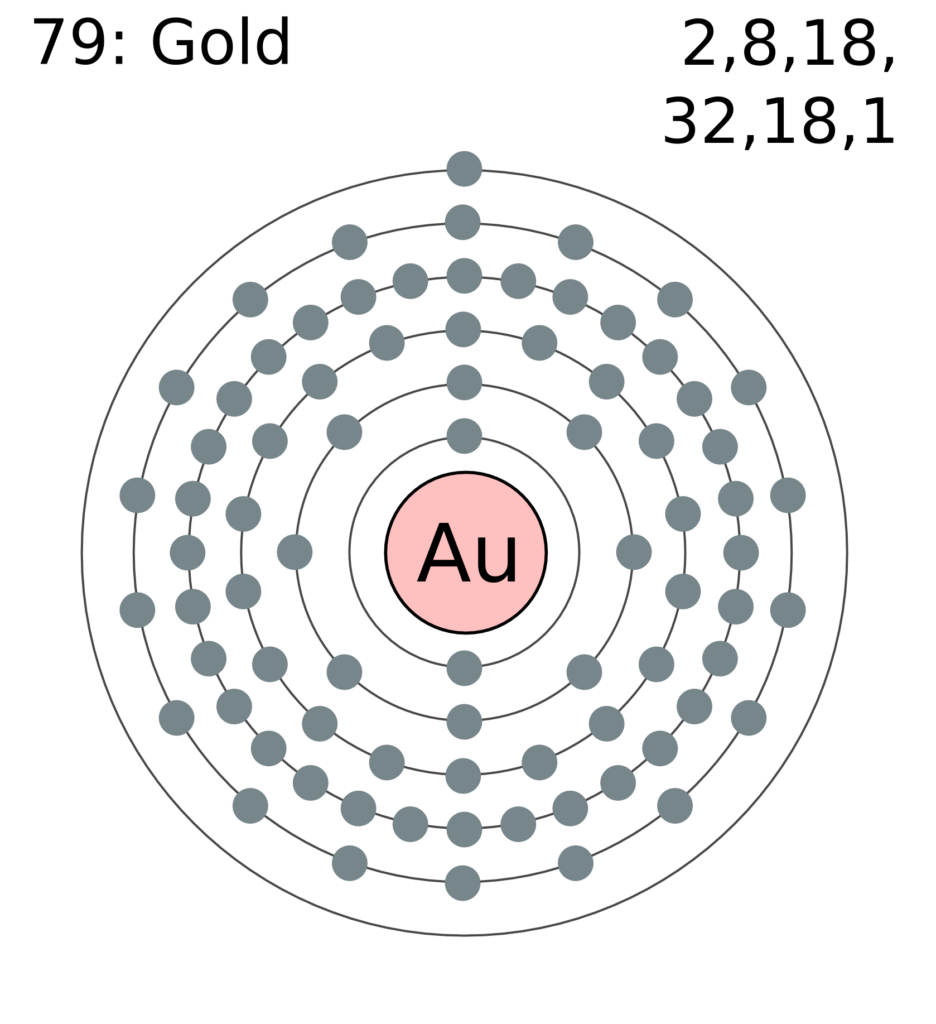
The most reactive nonmetals are the halogens, e.g., #"F"# and #"Cl"#. Nonmetals tend to attract additional valence electrons to form either ionic or covalent bonds. They need to lose only one or two valence electrons to form positive ions with a noble gas configuration. The most reactive metals are those from Groups 1 and 2. Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell with a noble gas electron configuration ending in #ns^2 np^6#. The electrical configuration and number of valence electron of elements in the second period are tabulated below.Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table. Valence Electrons of Elements in Periodic Table Hence, we can rapidly calculate the total number of valence electrons in an element by writing down its electrical configuration. You can quickly get an idea of how many electrons are in the outermost shell by looking at the electron configuration. Electronic configuration refers to the specific configuration of these orbitals occupied by electrons. The orbits of electrons within the shells are known as atomic orbitals. The rule is valid for elements in groups 3–12 that are in transition or an inner transition.īy the Use of Element Electronic Configuration Nevertheless, this only applies to the elements of the two main groups (groups 1 and 2) and groups (13-18). Note: The number of shells is indicated by the period, whereas the number of valence electron in the outermost shell is indicated by the group. On the other hand, going from left to right throughout a period results in an increase of one valence electron. The number of valence electron remains constant even when the number of shells expands as we move from the top to the bottom of a group. Here, we just look up the element in the periodic table to get its specific location. In order to count the valence electron of an element, this is the most used technique. The number of valence electrons in an element may be determined in two methods. How to find Valence electrons of the element?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
